

The Green Research Group works at the interface of Chemistry, Biology and Physics and thereby provides extensive training and collaborative opportunities for students in our program. Our group employs organic and inorganic synthetic methods along with spectroscopic/analytical techniques (electrochemistry, XRD, NMR, UV-vis and others) to target current challenges in biomedicine and chemical industry. We collaboratively complement our projects with cellular biology, physical inorganic methods (EPR, advanced NMR, determination of binding constants, and computational methods), and studies carried out with animal models (murine to date). Our current research focus includes the development of small molecules that can target diseases that involve oxidative stress and/or metal-ion misregulation, such as Alzheimer’s. We are also developing a new class of metal-based complexes that can be used to study and ultimately optimize C-C and C-H transformations in organic chemistry as well as new contrast agents derived from paramagnetic metal ions.
The TCU Chemistry & Biochemistry Graduate Program is currently accepting applications for new graduate students! Find out more here.
Research Focus: Develop Antioxidant Small Molecules to treat Neurodegeneration and diseases of the eye
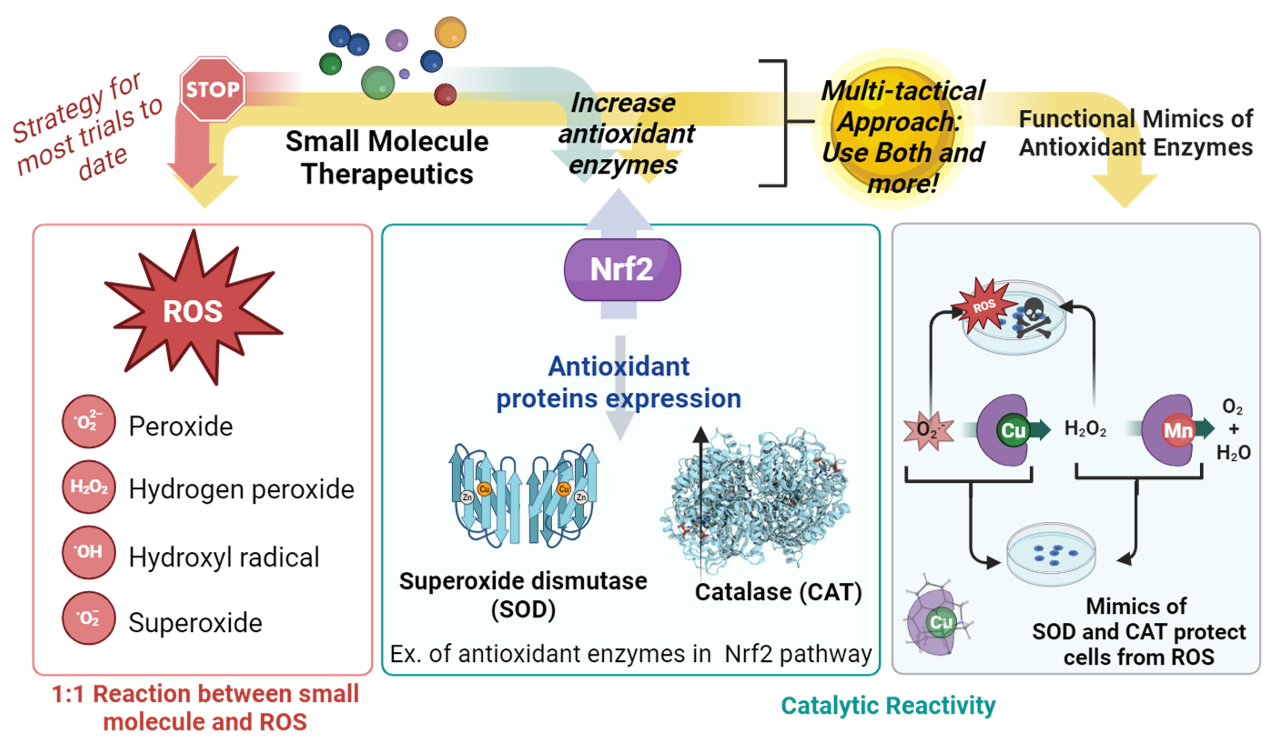
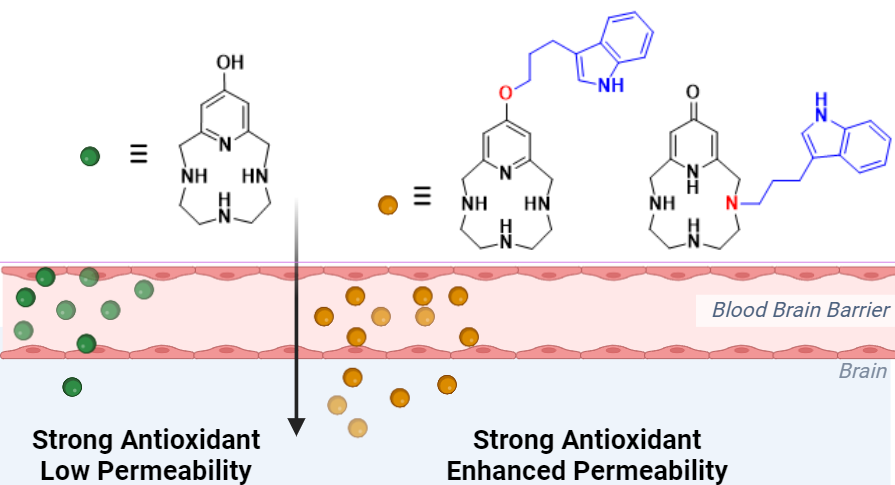

Inorg. Chem. 2024, 63, 50, 23544–23553. What makes for a potent and efficient SOD mimic? We have found that balancing the Cu(II) vs. Cu(I) stability is key for this mechanism. Read more here!

